For menn som lider av hårtap, varierer årsakene fra arvelige egenskaper til underliggende medisinske tilstander til livsstilsvalg og mer. Skulle du merke en avtagende hårgrense eller tynnere hår ved kronen på hodet, er det første du må huske på at du er langt fra alene. Faktisk tyder studier på at omtrent 85% av mennene opplever betydelig tynning av hår i en alder av 50 år.
Hva er hårtap?
Som det viser seg, opplever både menn og kvinner hårtap hver dag (omtrent 100 hår, faktisk). Men når du har et sunt hår, blir gamle hår raskt erstattet av nye. Som et resultat merker du ikke tynning av hår ved kronen eller andre tegn på mannlig skallethet. Naturligvis refererer vi ikke til denne prosessen når vi bruker begrepet “hårtap” i generell forstand. Det er fordi faktisk hårtap oppstår når syklusen av gjenvekst blir forstyrret og follikelen blir skadet uten reparasjon.
Et annet ord for hårtap er alopecia, og det kan ha forskjellige former, som bare påvirker hodebunnen din eller noen ganger fjerner alt håret fra kroppen din. Den mest typiske typen alopeci blant menn er mannlig skallethet og utgjør omtrent 95% av hårtapet.
De beste årsakene til håravfall
For de fleste voksne menn er hårtap mer eller mindre et faktum i livet. Det er fordi genetikk spiller den største rollen og med ganske stor margin. Imidlertid, ikke ta det for å bety at det ikke er andre faktorer å vurdere. Her er 6 vanlige hårtap hos menn.
1. Arvelige egenskaper
Hva forårsaker skallethet? Du spør. Svaret er oftest dine forfedre. Faktisk lover en familiehistorie med hårtap ikke godt for folliklene dine når du blir eldre.
2. Underliggende medisinske forhold
Enkelte medisinske tilstander som ringorm eller skjoldbruskkjertelsykdom kan forstyrre din hormonbalanse og føre til plutselig hårtap som et resultat. Noen sykdommer (for eksempel spesifikke typer lupus) kan skape arrdannelse i hodebunnen, og dermed muliggjøre permanent håravfall.
Tilstander som diabetes kan skade immunsystemet, noe som kan føre til hårtap som alopecia areata. Det er også en hårtrekkforstyrrelse kjent som trichotillomania, som kan gi midlertidig skalling (antar at folliklene ikke er skadet).
3. Medisiner og behandlinger
Enkelte typer medisiner og behandlinger inkluderer hårtap som en potensiell bivirkning. Det inkluderer medisiner for sykdommer eller tilstander som kreft, høyt blodtrykk, leddgikt, depresjon og hjerteproblemer.
4. Ekstrem stress
I kjølvannet av et fysisk sjokk eller en traumatisk hendelse opplever mange mennesker midlertidig hårtap. Dette skyldes hovedsakelig forholdet mellom stress og hormonproduksjon.
5. Livsstilsvalg
Kosthold som mangler proteiner, jern og viktige vitaminer og næringsstoffer har vært knyttet til tynnhår, mens røyking har blitt påstått for å fremskynde symptomene på mannlig skallethet. Ifølge noen studier kan både overdreven vektøkning og raskt vekttap gi visse mangler og dermed forårsake hårtap.
Forebygging av hårtap
Siden de fleste årsakene til hårtap er genetiske, kan man se på det som en uunngåelig tilstand. Selv om det til en viss grad er sant, er det tiltak du kan ta for å bremse prosessen med alopecia. Det er også viktig å huske at ikke alt hårtap er et resultat av en arvelig egenskap, noe som gjør visse tiltak mye viktigere. Her er hva du kan gjøre for å forhindre hårtap.
1. Bruk behandling for håravfall
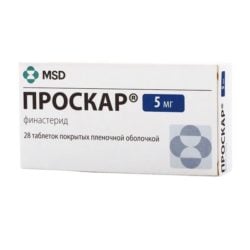
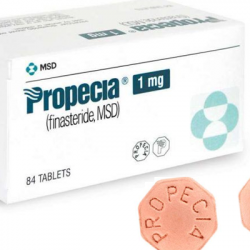
De to mest populære behandlingene for hårtap (også kjent som hårvekstbehandlinger) er Minoxidil og Finasteride. Du kjenner sannsynligvis disse medisinene under populære merkenavn som Rogaine (Minoxidil) og Propecia (Finasteride).
Finasteride krever en ganske lett å få tak i resept og kommer i pilleform. Selv om begge disse behandlingene fungerer annerledes, har de vært direkte knyttet til å forebygge hårtap og til og med fremme gjenvekst hos visse brukere.
Hvis du tar Finasteride, må du være oppmerksom på at det kan ta tre måneder (om ikke lenger) før du ser resultater. Det er også en foreskrevet medisin, og det betyr at du kan oppleve potensielle bivirkninger som depresjon, kløe, impotens og mer.
Hvor lang tid tar det før behandling av hårtap fungerer?
Finasterid og minoksidil tar vanligvis tre til fire måneder daglig bruk for å begynne å se synlige endringer. Det kan ta opptil et år å se maksimale resultater. Så lenge du ikke opplever bivirkninger, er det best å fortsette medisinen i ett helt år før du bestemmer deg for om medisinen gjør alt den kan for deg. Som alle medisiner vil finasterid og minoksidil fungere annerledes for forskjellige mennesker. For å opprettholde fordelene, må du fortsette å bruke hårtapmedisiner. Hvis du stopper, kan hårtapet komme tilbake.
Viser alle 2 resultatene